Molar entropy and absolute entropy4/19/2024  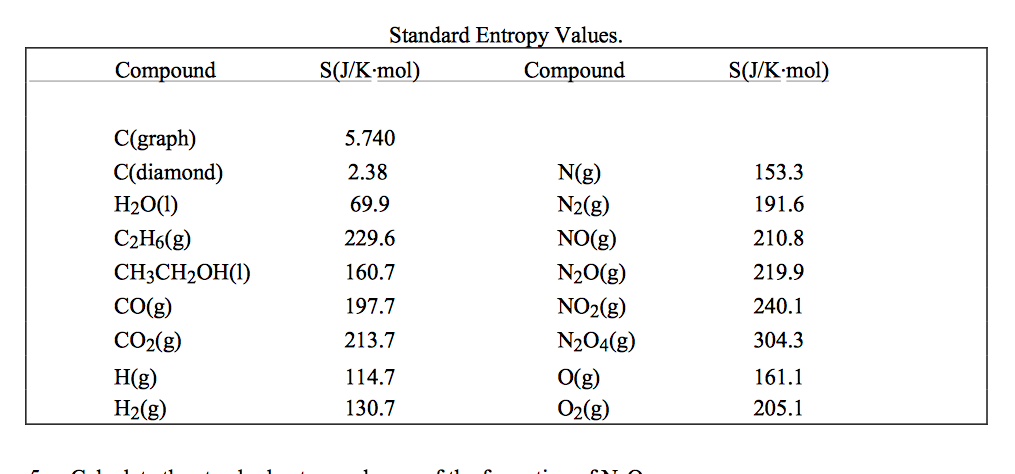
A molecule with more atoms will, in general, have a more degrees of freedom to take up energy, increasing its number of available microstates and entropy.\hspace-.6pt\raise1. The absolute or standard entropy of substances can. As the temperature of the substance increases, its entropy increases because of an increase in molecular motion. Standard absolute entropy refers to the absolute entropy of a substance in its standard state (that is, its state at 298.15 K and atmospheric pressure). Therefore, the entropy of a pure crystalline substance at absolute zero is defined to be equal to zero. the entropy of a pure substance at 298 K and 1 atm pressure). All molecular motion ceases at absolute zero (0 K) ( 0 K). Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. In equations, entropy is usually denoted by the letter S and has units of joules per kelvin (JK 1) or kgm 2 s 2 K 1. The entropy of a substance has an absolute value of 0 entropy at 0 K. Standard Molar Entropy, S Larger, more complex molecules have higher standard molar enthalpy values than smaller or simpler molecules. Thus, entropy has the units of energy unit per Kelvin, J K -1. Entropy is the amount of energy transferred divided by the temperature at which the process takes place. Entropy contained in a system, say in a mole of a pure substance, is a theoretical quantity that takes account of all heat transferred to it since the lowest atainable temperature, 0 K. When a system receives an amount of energy q at a constant temperature, T, the entropy increase D S is defined by the following equation. It is an extensive property of a thermodynamic system, which means its value changes depending on the amount of matter that is present. By definition, the change in entropy can be evaluated by measuring the amount of energy transferred. The third law derived from experiments looking at the behavior of heat capacities and entropy at lower and lower temperatures. As the temperature increases, the entropy of the atoms in the lattice increase. 
The position of the atoms or molecules in the crystal would be perfectly defined. The third law, like the other laws of thermodynamics, is derived from empirical observations made by scientists studying the behavior of thermodynamic systems. The entropy of a pure crystalline substance at absolute zero (i.e. The same is true for the number of atoms in a molecule. Entropy is the measure of the disorder of a system. The Third Law and calculation of absolute entropies. Absolute standard entropy, S 298, represents thermodynamic data of special significance, forging the link between enthalpy and Gibbs energy, which is the. Because the entropy of a substance depends on the amount of substance, the pressure, and the temperature, it is convenient to describe the entropy of a substance in terms of S m °, its standard molar entropy, i.e., as the entropy of 1 mol of substance at the standard pressure of 1 atm (101.3 kPa) and given temperature. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
